ST05
Separation of homogeneous solid - liquid mixtures
| Aim:
To introduce distillation as one of the most important methods
for separating homogeneous solid - liquid and liquid -
liquid mixtures (solutions). |
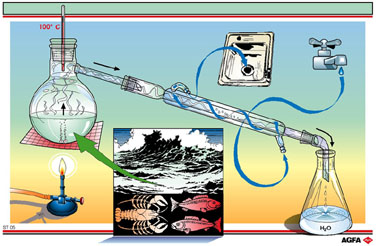
By ‘distillation’ we mean: a separation process in which one of the components is made to boil, and the vapour is separated and made (by cooling) to condense separately. An example is the distillation of seawater as shown on illustration ST05. This is the separation of non-volatile substances (the dissolved salts) from a volatile liquid (water).
The
seawater is warmed in a round-bottomed flask until it boils, whereby water
vapour is formed. This vapour is condensed in a Liebig condenser. The
resultant water droplets, the distillate, are collected in a recipient.
The originally dissolved salts remain in the flask (the residue). This
is one of the possible ways of obtaining drinking water from seawater
(see also ST06 and ST09).
Mixtures of two or more volatile components can also be
separated by distillation.
The theory of this type of distillation is explained in the text on illustration
ST07.