>Past
President's Column
>PEDECIBA
>Scientific and Technical Information
>Macromolecular
Nomenclature and Terminology
>Aligning
the Red and Blue Books
>IUPAC
News
>Awards
>IUPAC
Projects
>Highlights
from PAC
>Provisional
Recommendations
>New
Books
>Reports
from Conferences
>Conference
Announcements
>Conference
Calendar
Chemistry International
Vol. 24, No. 6
November 2002
IUPAC Projects
New SIT Software Makes Speciation Calculations Easier
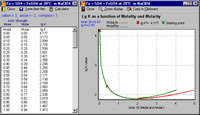
Metal ion speciation in environmental systems is a hot topic because speciation is strongly linked to bioavailability. For labile systems, the speciation can be determined by equilibrium modelling calculations to give the well known "speciation diagram," a plot of composition (species concentration) vs. pH or composition vs. metal/ligand ratio.
Speciation calculations require values for the stability constants of all the species that may form from the system components. These values must be valid at the ionic strength of the medium concerned. Here is a major problem: the vast majority of equilibrium constants have been determined at a fixed ionic strength that is typically much higher (ca. 0.1-1.0 M) than applies in freshwater environmental systems (<0.02 M).
To correct the stability constants to the required ionic strength one must be able to calculate the activity coefficients of all species in the equilibrium reaction. These corrections may be quite large (e.g., log K1 for the reaction Fe3+ + nta3- is 16.26 for I=0.10 M NaClO4 and the value calculated for I=0.001 M is 17.89).
The second problem is that not all equations for activity coefficient calculations (e.g., Debye-Hückel and Davies [eqtn. 1]) are valid to such high ionic strengths:
log gI = -AzI2{|1/2/(1+|1/2)-0.3|} (1)
Specific Interaction Theory, or SIT (and Pitzer parameters), provides a valid tool for calculating stability constants over a wide range of ionic strengths. The calculations are not trivial; they require a database of ionic coefficients, eM,X, for each ion-ion interaction:
log gM = -Azi2{|1/2/(1+1.5|1/2)+e(M,X)mX} (2)
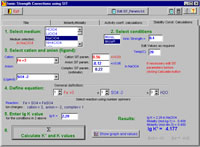
Now for the easy and exciting part! An IUPAC project, undertaken by L. D. Pettit of Academic Software, has produced the SIT program that will calculate:
- ionic activity coefficients in a user-specified medium, up to 5.0 molal
- stability constants in the range 0-5.0 m, based on published values at a single ionic strength
- molarity from molality, and vice versa
This is an enormous asset for those involved in speciation calculations. It is a user-friendly program that is now in the public domain at <www.iupac.org/projects/ 2000/2000-003-1-500.html>. Interested persons are invited to test the SIT program and send comments to the author. The program includes a comprehensive, but not exhaustive, file of SIT parameters. The file can be edited, but if users have access to reliable SIT parameters that are not included, please send these to the author at <www.acadsoft.co.uk> so that the release version of the program can be updated for all to benefit.
The SIT program can be readily applied to industrial processes for which stability constants may be required at very high ionic strengths.
Two other IUPAC products will assist in the understanding and application of stability constants. The IUPAC Stability Constant database, SC-database1 should be a starting point for stability constant data. It links data directly to the program SPECIES that will calculate speciation curves. The HELP files in the database provide definitions and information about temperature, ionic strength, and solvent effects. For a much wider discourse, the program Sol-Eq2 explains the principles of solution equilibrium and explores many applications in environmental, biological, and industrial systems. It includes a primer on how to do your own speciation calculations.
- SC-database, Stability Constants Database; IUPAC, Academic Software. 2001.
- Sol-Eq. Solution equilibria: Principles and Applications. Academic Software. 2001.
Both of these programs are available to order via <www.acasoft.co.uk>.
Reviewed by Kip Powell, vice president, Analytical Chemistry Division.
![]()
News
and Notices - Organizations and People
- Standing Committees
Divisions
- Projects - Reports
- Publications - Symposia
- AMP - Links
Page last modified 31 October 2002.
Copyright © 1997-2002 International Union of Pure and Applied Chemistry.
Questions or comments about IUPAC, please
contact the Secretariat.
Questions regarding the website, please contact [email protected]