Vol.
29 No. 5
September-October 2007
Wet Surface Vibrational Spectroscopy Experiments
In recent years, there have been important developments in vibrational spectroscopic techniques applicable to studies of the wet interfaces which surround us in nature and in technology. Methods that can provide vibrational spectroscopic information about monolayers at surfaces are powerful tools in revealing the molecular basis of the observed macroscopic behavior of such systems. Surface enhanced Raman spectroscopy (SERS), which gives remarkable enhancement of typically weak Raman scattering, was at first observed from silver and gold electrodes. SERS is now more widely observed in other contexts and is even used for single molecule detection.1 Surface enhanced infrared absorption spectroscopy (SEIRAS) is a related technique which gives enhancements of infrared absorption and is used mainly in electrochemical contexts.2 The deposition of thin films of colloid-like particles on prisms used in attenuated total reflection infrared spectroscopy (ATRIRS) provides signal enhancements due to the high surface area and facilitates wet adsorption studies of diverse materials.3 Presently, the most exciting surface-sensitive spectroscopic technique is vibrational sum frequency spectroscopy (VSFS).4 This nonlinear optical technique provides vibrational spectra of molecules at “buried” interfaces and is highly surface selective due to symmetry changes at interfaces.
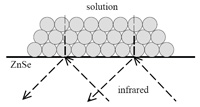 |
Internal reflection of infrared radiation and penetration of evanescent wave through wet particle film in ATRIRS. (Reproduced from reference 3 with
permission.) |
The aim of this project is to promote the application of ATRIRS, SEIRAS, and SERS to problems in interfacial chemistry by selecting, testing, and disseminating a collection of experiments suitable for undergraduate teaching laboratories, which are able to be performed with relatively inexpensive equipment. Undergraduate experiments in interfacial chemistry are presently dominated by measurements of macroscopic quantities such as surface tension and amount adsorbed when increasingly spectroscopic and microscopic data are presented in the corresponding lectures. This project takes a lead in encouraging a more modern molecular approach to interfacial physical chemistry teaching experiments by compiling and testing a series of appealing experiments which can be readily carried out in undergraduate laboratories. The relatively high cost and technical sophistication of VSFS precluded its inclusion in this project.
This project brings together leading physical chemists in the fields of attenuated total reflection infrared spectroscopy (ATRIRS), surface enhanced infrared spectroscopy (SEIRAS), and surface enhanced Raman spectroscopy (SERS), to select and test practicable experiments which can be carried out in teaching situations throughout the world. Both SERS and SEIRAS employ finely divided metal surfaces while the ATRIRS particle film approach can be applied to any particulate solid. All of the chosen surface spectroscopies are applicable to solid/aqueous interfaces that are of considerable interest in studies of natural and technological systems. Examples include the study of adsorbed electrode intermediates and the investigation of adsorbed collectors used as flotation agents for mineral extraction.
The experiments will include the basis of the different spectroscopic techniques, the preparation of surfaces to which the techniques can be applied, and several experimental systems as examples. Most undergraduates have access to infrared spectrometers in teaching laboratories so that the ATRIRS and SEIRAS experiments are expected to find more widespread use. SERS experiments have been included because Raman instruments are decreasing in cost and are now more frequently found in undergraduate laboratories.
The experiments will be tested and refined in their laboratories of origin, followed by testing in university undergraduate laboratories elsewhere under normal laboratory conditions. The project outcomes will be disseminated through the IUPAC website which will provide a means of assessing the uptake of the developed material.
References
1. Z.Q. Tian and Bin Ren, Ann. Rev. Phys. Chem. 55, 197–229 (2004)
2. M. Osawa, Top. Appl. Phys. 81, 163–187 (2001)
3. A.J. McQuillan, Adv. Mater. 13, 1034–1038 (2001)
4. Y.R. Shen, Pure Appl. Chem. 73, 1589–1598 (2001)
For more information and comments, contact the Task Group Chair Jim McQuillan <[email protected]>.
www.iupac.org/projects/2006/2006-050-3-100.html
Page
last modified 18 September 2007.
Copyright © 2003-2007 International Union of Pure and Applied Chemistry.
Questions regarding the website, please contact [email protected] |