|
|
Vol.
35 No. 4
July-August 2013
by Takashi Ushikubo
In Japan, Green and Sustainable Chemistry (GSC) is defined as innovations and advances in chemical technologies that can improve human and environmental health. The Green Chemistry Initiative, which was formed in September 1998 and comprised companies, universities, and the government, was intended to encourage technologies that could be used in sustainable development. Out of this initiative, the Green and Sustainable Chemistry Network (GSCN) was born in March 2000.
Although we did not have the term “GSC” in the 1960s, the research and development that form the foundation of industrial ecology were already taking place. For example, it was during this decade that mercury cell electrolysis was replaced by the ion exchange membrane method for the production of sodium hydroxide. The introduction of this environmentally benign process had been promoted by the Association for the Progress of New Chemistry, which was the forerunner to the Japan Association for Chemical Innovation (JACI). The GSCN, which is managed by JACI, is the center of a network that actively promotes collaboration in research and development through information exchange, communication, education, and international activities.
This article surveys recent progress in GSC technologies in Japan and the expectations for future innovation. In addition, it discusses the potential contribution of GSC toward reviving cities, towns, and the industries damaged by the Great East Japan Earthquake.
One of the activities of GSCN is the GSC Awards, started in 2001. GSC Awards are granted to individuals, groups, or companies that have greatly promoted GSC through their research, development, or commercialization.1 These awards are given by Japan’s Minister of Economy, Trade, and Industry; Minister of the Environment; or Minister of Education, Sports, Culture, Science, and Technology, depending on the particular achievement. By reviewing the winners of the GSC Awards, one gets a clear picture of the environmentally benign processes that have been commercialized in Japan in the past decade.
Polycarbonate is one of the most important and versatile engineering plastics. With superior transparency, impact resistance, and heat resistance, it is used in optical discs, home electronics, automobile parts, office equipment, and so on. About four million tons of polycarbonate is produced annually worldwide, with production expected to grow in future years. Almost all polycarbonate was manufactured through a process in which CO and chlorine are combined to form phosgene, a highly toxic gas, as an intermediate material. The phosgene process has a number of disadvantages, including the risk of environmental harm. Many attempts have been made to overcome the inherent risks in the phosgene process, but with little success until recently.
Asahi Kasei Corporation succeeded in commercializing the first non-phosgene polycarbonate production process in the world. The figure below compares this new method with the conventional phosgene process. This technology uses ethylene oxide and CO2, that is the by-product of ethylene oxide synthesis. Ethylene carbonate is synthesized by the reaction between ethylene oxide and CO2, and ethylene carbonate is converted to diphenyl carbonate via dimethyl carbonate. Furthermore, diphenyl carbonate reacts with bisphenol-A to produce high-quality polycarbonate. Also, high-purity monoethylene glycol is co-produced. This technology successfully incorporated CO2, which has a low chemical activity, into the polycarbonate main chain. In addition, this new process does not use methylene chloride as a solvent. This novel technology totally eliminates the problems of the phosgene process, while maintaining high production yields and conserving resources and energy. The new process reduces CO2 emissions by 0.173kg per one kg of product polycarbonate.
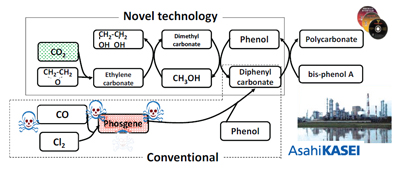 |
| Novel non-phosgene polycarbonate production
process using by-product CO2 as a starting material. |
ε-Caprolactam is an important intermediate used in the production of Nylon 6 fibers and resins, the worldwide production of which is estimated at about 3.8 million tons annually. Nylon 6 fibers and resins are used in a wide range of applications including clothes, automobiles, electric or electronic devices, as well as lapping films for foods. Almost all ε-caprolactam is produced through the Beckmann rearrangement of cyclohexanone oxime, and a large amounts of ammonium sulfate is co-produced, at least 1.6 times of caprolactam by weight, because highly concentrated sulfuric acid is used for the reaction promoter and the neutralization by ammonia is necessary in the process.
Sumitomo Chemical Company succeeded in commercializing the novel catalytic Beckmann rearrangement reaction proceeded under vapor phase conditions without ammonium sulfate co-formation. This process is conducted with a high-silica MFI zeolite catalyst instead of sulfuric acid. And an effective purification technology was developed to obtain a high-purity product with a reduction in the consumption of resources and energy. In order to carry out the production of ε-caprolactam without any ammonium sulfate production, Sumitomo Chemical commercialized a combined process in 2003, which involves an initial ammoximation step for obtaining cyclohexanone oxime, developed by EniChem (Italy), and a secondary step involving a vapor phase Beckmann rearrangement (see figure below).
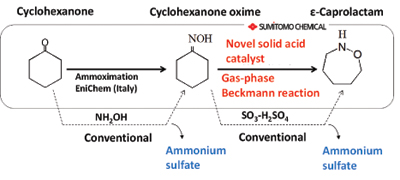 |
| Novel ε-caprolactam production without ammonium sulfate formation. |
Mono-ethylene glycol (MEG; HOCH2CH2OH) is used as an antifreeze and as a raw material for the production of polyester fibers and resins, mainly PET. A new process was developed by Mitsubishi Chemical Corporation for the production of mono-ethylene glycol to meet its growing demand in the world. While the MEG selectivity of the conventional non-catalyzed process is around 89 percent under typical conditions, the new process exceeds 99 percent. The key to the high selectivity is the two-step synthesis via ethylene carbonate, as in the figure above. The first step reaction is catalyzed by phosphonium salts and generates ethylene carbonate as an intermediate. This is followed by hydrolysis of ethylene carbonate in the second step. The hydrolysis is carried out under almost stoichiometric condition, although the conventional non-catalyzed hydrolysis is accomplished by the higher H2O/ethylene oxide molar ratio. Several commercial plants utilizing the new process are already in operation throughout the world. This greener technology saves resources and the energy and reduces the amount of wastewater and CO2 production.
In the early stages of Green and Sustainable Chemistry promotion in Japan, the GSCN focused on developing safer industrial technology, decreasing the environmental burden of industrial production (i.e., carbon dioxide, waste, and harmful byproducts), and on establishing a new philosophy and methodology for chemical research. Guidance from the Organization for Economic Cooperation and Development was helpful to Japan as it sought to improve the safety of chemicals and decrease pollution. A number of laws have been passed to reduce pollution and to promote recycling.
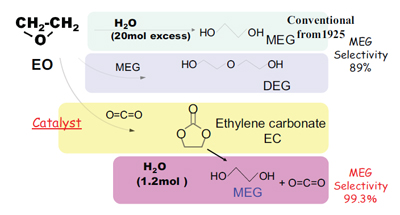 |
| Ethylene glycol synthesis process using novel
homogeneous catalyst. |
Current GSC technologies contribute to sustainability, not only by decreasing the environmental impact of industrial processes, but also by increasing the prosperity of society. In order to expand GSC, the Ministry of Economy, Trade and Industry released the Technology Strategy Map on GSC in 2007. The 2010 issue of the map gave an overview of 153 GSC technologies that were related to the sustainability of energy, environment, natural resources, and a prosperous society. The Ministry is strongly promoting national research projects having to do with Green Innovation and Life Innovation.
Universities and national laboratories have been collaborating with companies to develop fundamental science and technologies, such as catalysts, super-critical fluids, analysis, and computer chemistry. Also, the chemical industry has developed innovative sustainable materials for products such as photovoltaic power generation, hybrid/electric vehicles, water treatment, and high-function fiber, that accelerate the challenges in energy, the environment, and healthcare. In modern cars, for instance, innovative polymer materials reduce the weight and thereby increase the cars’ energy efficiency.
The Great East Japan Earthquake of 11 March 2011 devastated certain areas of the country, especially the Tohoku district, and resulted in numerous long-term problems such as a diminished energy supply, extensive cleanup of land and water polluted by radioactive materials, and the need to rebuild industries and increase employment. Green and sustainable chemistry is expected to play an important role in the recovery effort. JACI has started the New Chemical Technology Research Encouragement Award, including an Award for Earthquake Disaster Recovery, to support the activities of young researchers engaged in researching new chemistry. In 2011, the following three researchers won the award for their unique ideas:
- Yoshitaka Takagai: Development of Speedy Radioactive Strontium Analysis Aystem and Its Application to Soil Monitoring Concerning Tokyo Electric Power Co.,Inc, Fukushima Daiichi Nuclear Power Plant Accident
- Shunsuke Kurosawa: Development of a Novel Scintillator for Radiation Monitor in the River
- Masami Hatayama: Phytoremediation of Heavy Metals and Metalloids Derived from Tsunami Sediments
In order to promote GSC for the future, JACI has been educating the public, especially younger students, about the broader benefits of chemistry for the planet and for society. JACI published the introductory textbook Chemistry and Environment: An Introduction to Green Chemistry for college and high school students. The first edition was published in April 2002 and has been used as a textbook at several colleges.
 |
| The contribution of chemical technologies toward
a sustainable society. |
In addition, some aspects of GSC can be taught through micro-scale chemistry experiments, which are themselves environmentally benign. Kazuko Ogino of Tohoku University has been developing, promoting, and disseminating micro-scale chemistry experiments for some time. Ogino won the Foundation of Asian Chemical Society Distinguished Contribution to Chemical Education Award in 2011 and the GSC Award in 2007. Chemical companies and JACI are supporting such activities in Japan by helping the public to understand the features and benefits of micro-scale chemistry experiments.
- http://www.gscn.net/awardsE/index.html
- K. Komiya, S. Fukuoka, M. Aminaka, K. Hasegawa, H. Machiya, H. Okamoto, T. Watanabe, H. Yoneda, I. Fukawa, T. Dozono, Green Chemistry, ACS Symposium series 626, 20, 1994; S. Fukuoka, I. Fukawa, M. Kawamura, K. Komiya, M. Tojo, H. Hachiya, K. Hasagawad, M. Aminaka, H. Okamoto, S. Konno, Green Chemistry, 5, 497, 2003.; S. Fukuoka, I. Fukawa, M. Tojo, K. Oonishi, H. Hachiya, M. Aminaka, K. Hasegawa, K. Komiya, Catalysis Surveys from Asia, 14, 146, 2010.
- H. Ichihashi, M. Ishida, A. Shiga, M. Kitamura, T. Suzuki, K. Suenobu and K. Sugita, Catalysis Surveys from Asia, 7, 261, 2003.
- K. Kazuki, Catalysis Surveys from Asia, 14, 111, 2010.
- http://www.meti.go.jp/english/policy/economy/growth/outline20100618.pdf
Professor Takashi USHIKUBO <[email protected]> works with the Japan Association for Chemical Innovation, in Tokyo, Japan.
Page
last modified 29 August 2013.
Copyright © 2003-2013 International Union of Pure and Applied Chemistry.
Questions regarding the website, please contact [email protected]
|