|
|
Vol.
35 No. 3
May-June 2013
by Bernardo Herold
International organizations, such as the European Union, make extensive use of IUPAC nomenclature to organize and maintain huge databases of chemical substances. However, this task can be quite difficult when the data in question is multilingual, as is the case with the EU.
 |
Although English is the official language of IUPAC and it therefore does not make any recommendations on how to name chemical substances in other languages, some of the most important nomenclature books or recommendations have been translated into other languages. A list of these translations compiled by Dr. Gerard P. Moss can be found at <www.chem.qmul.ac.uk/iupac/
bibliog/books.html>. Such translations are of great value to those in charge of translating the names of substances at international organizations. If there are no translated nomenclature rules in a given language, the task of translating, name by name, in a database with tens of thousands of entries, becomes very difficult and arbitrary. There is a great risk that such translated terms will be inconsistent or
ambiguous.
The publication of a translation of IUPAC Nomenclature Recommendations is subject to the following rules, found at <www.iupac.org/home/publications/technical-reports/procedure-for-publication.html>:
- Republication or reproduction of any Recommendation or Technical Report or its storage and/or dissemination by electronic means. No formal IUPAC permission is needed on condition that an acknowledgment, with full reference to the source along with use of the copyright symbol ©,
the name of IUPAC, and the year of publication are prominently visible.
- Publication of a translation into another language is subject to the additional condition of prior approval from the relevant IUPAC National Adhering Organization.
These rules, however, do not explicitly address cases in which two or more countries use the same language. The Interdivisional Committee on Terminology and Symbols filled this gap by insisting that in such cases the translation must be approved by all National Adhering Organizations (NAOs) relevant to that language. However, there are cases where the chemical communities of the involved countries had little contact with each other during the period in which chemical nomenclature developed and were subject to different cultural influences. This is the case for Brazil and Portugal. During the first half of the 20th century, the dominant foreign cultural influence on the chemists in Portugal came from France, whereas, in Brazil, German-speaking immigrants and Brazilian chemists trained in the USA left their footprints on the way chemical nomenclature was adapted to the Portuguese language. The Portuguese translations of IUPAC nomenclature books obtained the approval of both concerned NAOs. The translator teams had members of both countries, who collaborated successfully in minimizing the differences between both versions. In the cases where no unification was possible, both the European and the Brazilian version were displayed in the translation. The following example, taken from the Portuguese translation of Carbohydrate Nomenclature, illustrates this practice:
The English name methyl α-D-glucopyranoside for
was translated in two versions as follows: α-D-glucopiranósido de metilo, (bras. α-D-glucopiranosídeo de metila). Fortunately, only one version was necessary for the great majority of the examples.
If IUPAC were to publish multilingual glossaries (either of substance names or other chemical terms), the same rules that apply to translations would have to be followed. In other words, such a glossary would require the approval of all involved NAOs, which could be a large number. For example, the 10 languages most important to the chemical literature (see table) are official languages in 80 countries, 26 of which are NAOs of IUPAC. In principle, they would each have to be asked for approval of the glossary.
Languagea
|
Number of countries with the same official language |
Number of IUPAC NAOs relevant to each language |
| Chinese |
3 |
1 |
| Czech |
1 |
1 |
| French |
29 |
5 |
| German |
6 |
6 |
| Italian |
2 |
2 |
| Japanese |
1 |
1 |
| Polish |
1 |
1 |
| Portuguese |
9 |
3 |
| Russian |
8 |
1 |
| Spanish (Castilian) |
20 |
5 |
| |
80 |
26 |
| a. The choice of languages and any deviation from official numbers in the second column have to be considered accidental and with no political intention. |
There are many technical aspects to the translation of nomenclature rules and names, which translators who have no training in chemistry, or chemists who are not trained as translators, may overlook. Some of the most important pitfalls for translators are presented below, but the most important recommendation is not to try to translate names of substances before the relevant rules have been translated.
The following is an example of the type of error made when attempting to translate substance names without knowing the translated rules:1
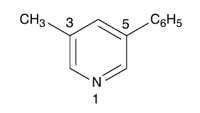 |
| 3-methyl-5-phenylpyridine, numbering in
accordance with the EN name. |
For the compound named “3-methyl-5-phenylpyridine” in English (EN), the following translation in Spanish (ES), Italian (IT), and Portuguese (PT) is wrong: “3-metil-5-fenilpiridina” (wrong alphabetical order of substituents) and “5-fenil-3-metilpiridina” (wrong numbering of ring). First, one has to draw the structure, based on the English name, order the substituents alphabetically in the target language and then number the ring atoms. The correct translation (ES, IT, PT) is thus “3-fenil-5-metilpiridina”.
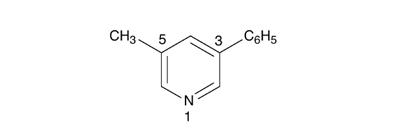 |
| 3-fenil-5-metilpiridina, numbering in accordance with the IUPAC rules translated in ES, IT, PT. |
It is curious to note that, if one derives the structural formula from any of the above quoted names, be they the correct or the wrong ones, one always arrives, nevertheless, at the same structural formula.
This example is only one of the many consequences of these three languages not using “ph” for the consonant “f”. Another case is the absence of the letter “y” in Italian, Portuguese, and Spanish, which does not allow any distinction between the translations of the retained names benzyl (C6H5–CH2) and benzil (C6H5–CO–CO– C6H5).
Another potential hazard for translators can be found in the new rules for coordination nomenclature (2005 edition of the Red Book). According to these new rules, the anion formerly called “tetrachloropalladate(2–)” is now named “tetrachloridopalladate(2–)”. But when translating into Portuguese, some have thought they should now use “tetracloridopaladato(2–)” instead of “tetracloropaladato(2–).” This typical error resulted from attempting to translate a name without having yet translated the relevant rules. The general rule in additive nomenclature applied to coordination compounds is that the (only sometimes modified) names of the ligands themselves are used as prefixes of the name of the central atom. The modifications recommended in English do, however, not always make sense in other languages: In English, the names of the anionic ligands ending with “ide”, “ate” and “ite” have to be changed to “ido”, “ato” and “ito”. In Portuguese and Spanish the last letter of the anion endings is already “o” (“eto”, “ato”, and “ito” in Portuguese and “uro”, “ato”, and “ito” in Spanish). Therefore, no modification has to be made, and thus, the part of the rules stating that the letter “e” should be changed to “o” does not need to be translated.
In the above example, the name of the anionic ligand is “cloreto” in Portuguese and “cloruro” in Spanish. Therefore, the name of the complex anion is “tetracloretopaladato(2–)” in Portuguese and “tetracloruropaladato(2–)” in Spanish. To use “clorido” in these languages as a ligand prefix is to completely misunderstand the intention of the IUPAC rules, which are intended to be as simple as possible and with as few exceptions as possible. In order to justify “clorido” as a prefix, one would have to introduce in the Portuguese and Spanish translations of the IUPAC rules a special exception (to modify “eto” to “ido”), which does not exist in the English original. Not only would it make the rules unnecessarily complicated, but it would introduce an obviously superfluous new term “clorido,” which had never been used before in these languages. Obviously, it is much simpler to tell a student that he or she has to use the unchanged name of the free ligand as the ligand prefix, than to have him or her memorize an extra rule.
As pointed out above for Italian, Spanish, and Portuguese, the fact that “y” is not part of the alphabet introduces potential ambiguity for those attempting to adapt the names “benzyl” and “benzil.” But the opposite can also be true: There are potential sources of ambiguity in English, which do not exist in many other languages. Such an example exists with the ending “ide”. It occurs in chemical nomenclature most often when designating an atom or group of atoms with a negative charge (i.e., an anion, like in “oxide”, “hydroxide”, “sulfide”, “sulfanide”, “methanide”, “azanide”, “phosphanide”, “nitride”, “fluoride”, “chloride”, and the other halides. In other cases, like in amides, imides, of acids, it does not signify a negative charge. The original meaning of the ending in Latin is “similar to.” As an example, “hominide,” derived from the Latin “homine” for man, means “similar to man.” When the root of the word is Greek as in “ anthropos” for man, the analogous term is “anthropoid.”
To prevent the misinterpretation of “actinide” as the monoanion of actinium, IUPAC decided recently to change the name of the group of elements “actinides” to “actinoids,” in spite of infringing on the classical rule in scientific terminology not to have a Greek ending with a Latin root. In this case, it was considered more important to remove the possible confusion between the group of elements and the anion. In other languages, such a change would not be necessary. I would not recommend changing the “actinídeos” in Portuguese to “actinóides,” because there is no risk of confusion with “actineto,” which would be the name of the monoanion derived from actinium. The same applies to Italian and Spanish, where the ending for the anion would be “uro”.
A problem relatively easy to solve is the inversion of the order of names in compositional nomenclature of inorganic compounds and functional class nomenclature of organic compounds (including the formerly so-called radico-functional nomenclature). Some simple examples are sufficient to illustrate the principle, which is common to at least four languages: French (FR), Italian (IT), Portuguese (PT), and Spanish (ES) in contrast with English (EN) and other languages:
NaCl: EN sodium chloride, ES cloruro de sodio, FR chlorure de sodium, IT cloruro di sodio, PT cloreto de sódio.
CH3COOC2H5: EN ethyl acetate, ES acetato de etilo, FR acétate d’éthyle, IT acetato di etile, PT acetato de etilo (bras. acetato de etila).
As can be verified in these two examples, in functional class nomenclature the words are separated by spaces in all five languages. This is not the case for German and some other Germanic languages:
For CH2(COOCH3)2, the translations of the English name dimethyl malonate are ES malonato de dimetilo FR malonate de diméthyle, IT malonato di dimetile, and PT malonato dimetílico, but in German (DE) it is Dimethylmalonat.
A slightly more complicated example in German is C(CH3)2(COOCH3)2: EN dimethyl dimethylmalonate, ES dimetilmalonato de dimetilo. FR diméthylmalonate de diméthyle, IT dimetilmalonato di dimetile, PT dimetilmalonato dimetílico, but the correct German translation is neither Dimethyldimethylmalonat nor Tetramethylmalonat, as one might suppose, but DE Dimethyl-2,2-dimethylmalonat.
As one may have noticed, there is an additional gender problem regarding the name of radicals or substituent groups between the Brazilian and European Portuguese versions. In European Portuguese, they are masculine, like “metilo”, “etilo”, “propilo”, etc. Once they cross the Atlantic and arrive in Brazil, they become feminine in “metila”, “etila”, “propila” etc. It has been impossible to reach an agreement. But should one really waste time on such a discussion? While Byzance was besieged by the Arabs, it is said that the church leaders were discussing the sex of angels. This might be a warning to overzealous multilingual nomenclaturists! The consequence of getting lost in such details might be that one forgets the most important aspect of translating nomenclature: First establish the rules in the target language before taking the second step, which is translating the names.
 |
Bernardo Jerosch Herold, born in 1933 in Lisbon to German-speaking parents, became a professor of organic chemistry at the Instituto Superior Técnico (IST) in Lisbon in 1962. His research in physical organic chemistry focused on radicals and ion pairs. Later in his career, he also taught and did research on the history of chemistry. Because of his thorough knowledge of both German and Portuguese, he was asked to translate the German text-book Organikum into Portuguese. This acquainted him with the problems of translating chemical nomenclature. In the 1980s, Herold coached for the Portuguese company Partex, as suppliers to the European Communities, a team of translators of 60 000 systematic names into the nine languages of the European Communities (at the time). He then felt the need to be in touch with the authors of the IUPAC nomenclature recommendations. In 1991, he became a member and later the secretary of Commission III.1 on Organic Nomenclature. Herold served the ICTNS Interdivisional Committee on Terminology, Nomenclature and Symbols as secretary from 2002 to 2011 and the Advisory Subcommittee of Division VIII Nomenclature and Structure Representation from 2002 until now. Herold co-authored the Portuguese translations of the 1993 Guide to IUPAC Nomenclature of Organic Compounds and the IUPAC and IUBMB Nomenclature of Carbohydrates. Presently he is working as a member of a team on the Portuguese translation of the 2005 Nomenclature of Inorganic Chemistry.
Herold's language skills developed from being bilingual and from being also taught French by his mother and in secondary school, as well as English. His French was improved by attending organic chemistry lectures at IST in the 1950s given in French by the French professor Pierre Laurent, as well as through industrial vacation training in France, dating a French-speaking girl, and attending a course on 19th Century French literature at the Alliance Française. His English was improved by attending a three-month English-language course in summer 1956 in London. Herold's German was updated by getting his doctoral degree 1961 in Heidelberg with Georg Wittig and marrying a German student. Later, his English was improved working in the English drama group Lisbon Players and by representing Portugal in various international organizations. As for his passive language knowledge, Herold picked up Spanish by reading Spanish books and traveling in Spain, and he once had some Italian lessons before traveling in Italy and can read Italian with the help of a dictionary. |
1. The author apologizes for not including examples of languages other than English, French, German, Italian, Portuguese, and Spanish, due to his lack of knowledge. For languages outside this group, there may be examples for translating problems of a different nature, which are not covered in this essay.
Page
last modified 14 May 2013.
Copyright © 2003-2013 International Union of Pure and Applied Chemistry.
Questions regarding the website, please contact [email protected]
|