|
|
Vol.
34 No. 3
May-June 2012
by Javier Garcia Martinez
and Rovani Sigamoney
Shortly after the UN declaration of 2011 as the International Year of Chemistry,1 IUPAC and UNESCO established a working group to devise a global project focused on promoting interest in chemistry among young people. Already in 2009, during the IUPAC General Assembly in Glasgow, it was suggested that this activity would be a collection of simple experiments related to water and that the results would be shared online. The central idea behind the project was that the kids would be the main actors of their own learning by having them leave the classroom for simple experiments related to daily activities, and then enabling them to share their experiences, pictures, and results on the web.
The various experiments were designed to require minimal equipment and resources, and the protocols drafted by an international team of educators.2 A dedicated website for the Global Experiment3 was created so that one could download the instructions for performing the experiments, upload the results, and view the data obtained by other students from around the world. The interactive site—available in English, French, Spanish, Russian, and Chinese—also served as a clearinghouse for teachers and affiliated centers. It was designed for young people to actively participate in IYC and learn about the relationship between water and many of the world’s current problems—from food shortages to climate change—and how chemistry plays a fundamental role in understanding and resolving these challenges.
 |
In the past, similar projects that focused on educating students about the role of water were highly informative and raised awareness. However, for this IYC project, it was decided that by encouraging students to conduct simple hands-on experiments, they would learn why water is so unique and the central role of chemistry in providing clean safe water. In particular, they would discover water’s chemical and physical properties, the importance of chemical measurement to test water quality, and how to purify it. By participating in the Global Experiment, students learned important chemical concepts such as salinity, solubility, and pH.
This initiative directly addressed key objectives of IYC, namely to increase public understanding about how chemistry provides solutions to contemporary challenges and to promote interest among young people in science and chemistry in particular.
When all of the results of the experiment were posted by students, an impressive map began to emerge that illustrated the contributions from all schools, colleges, or institutions involved around the world.
The experiments were designed so they could be performed with a minimum of resources and so that the level of sophistication could easily be increased. The protocols were provided with additional educational materials to enable teachers of every level to implement the experiment at the appropriate educational level of his/her class or group of students. To make the experiments as widely accessible as possible, the instructions were translated into 11 languages.
The four experiments comprising the Global Experiment were divided into two groups
Activities related to the measurement of water quality:
- Acidity: Students learned to take measurements of local water pH using various indicators. Next, they were taught to use techniques for checking the reliability of the results obtained.
- Salinity: The salinity experiment enabled students to measure the salt content of water samples by evaporation until the sample is dried to constant weight.
Activities related to water purification:
- Filtration: In this activity the students constructed a filtration unit and evaluated its efficiency and capacity using a sample of local water. As with other activities, the data generated contributed to a global map of the results.
- Distillation: During this activity, students investigated an alternative way of purifying water using solar energy and the process of distillation. Additionally, students had an opportunity to design and build their own distillation system.
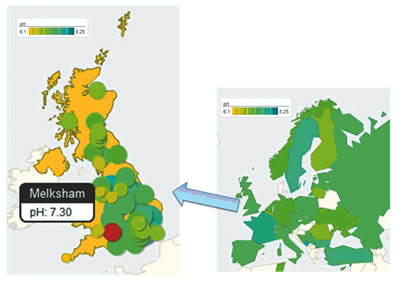 |
| Data collected by students are displayed on colorful maps that display the reported values, the location where the experiments were conducted, and the number of participants. |
These experiments were designed for a wide variety of educational settings. However, for the Global Experiment to be truly global, even children in regions with little to no resources needed to be able to participate. In order to include some students from these regions, the project task group arranged to send 150 free school kits, each consisting of 10 minilabs to 30 developing countries. This allowed the Global Experiment to reach 50 000 students who otherwise would not have been able to participate.
The project has afforded an enormous amount of quality water data from around the planet. Since launching the Global Experiment on World Water Day in March 2011 in South Africa, 128 330 students and 2354 teachers (as of 1 April 2012) from over 80 countries have shared their results on the website. However, the actual number of people who have taken part in the Global Experiment is much higher, since not all have shared their results online. In Brazil for example, it is estimated that more than 1.5 million students participated in this activity; support by the Brazilian government was instrumental in providing 30 000 kits for schools to participate. Similarly, in the United Kingdom where the Royal Society of Chemistry organized the Global Experiment Day on 22 June 2011, thousands of students participated in this activity. We may never know the exact number of students who participated, but the most important thing is that that through the Global Experiment thousands of young people around the world have discovered an interest in science and enjoyed doing simple experiments while learning about chemistry and water.
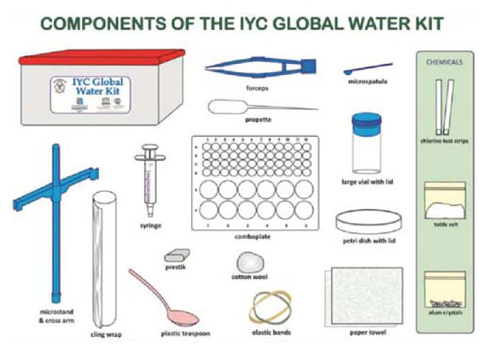 |
| Scheme of the minilabs experimental set, which also includes a scale and the necessary reagents to carry out all activities of the Global Experiment. |
Social media is a powerful tool embraced by the youth and which offers new opportunities and challenges for educators to engage their students. Central to the success of the Global Experiment was the use of social networks, which allowed students to shared experiences, photos, and results in real time through Facebook, Twitter, YouTube, or Flickr. Using these social tools, they were able to create their own communities of friends who shared their interest in chemistry.4
From the beginning, the Global Experiment was intended not just as an activity for youth. Instead, with the tools that social media provided, the experiment quickly evolved to become an activity by youth—a student-driven initiative allowing interaction through virtual communities. Both the students and the teachers are to be thanked, and each school that participated in this activity received a certificate of participation.
From the BBC to the Washington Post, a number of media outlets gave very favorable coverage of the Global Experiment, emphasizing its educational value and the fact that, overall, this was likely the largest chemistry experiment ever, given the huge number of participants. Perhaps just as important, the experiment showed how crowdsourcing can be used to acquire a large amount of scientific data that would otherwise be very difficult to obtain.
But how can we visualize the massive amount of data collected in a way that anyone can analyze, compare, and extract conclusions from the thousands of measurements reported? Recently, the Global Experiment was part of a competition organized by Visualizing.org, in which computer programmers worldwide were asked to develop software that could view, compare, and analyze the vast amount of information we gathered over the past year.5 In the spirit of the Global Experiment, this challenge was developed in a collaborative way so that from an initial design, programmers could create a more effective tool together than any individual programmer. The end result is an interactive map that displays all data sorted by type of experiment, the source of the water sample, and location.
On 14 March 2012, Colin Humphris and Javier Garcia Martinez, representing IUPAC, presented at the World Water Forum in Marseille, the results of this activity, the enormous amount of data reported, and the interest the experiment has generated for thousands of students. Although the Global Experiment has ended exactly one year after it began on World Water Day, IUPAC and UNESCO are starting to plan for the next activity that will promote interest in science and respect for the environment among young people.
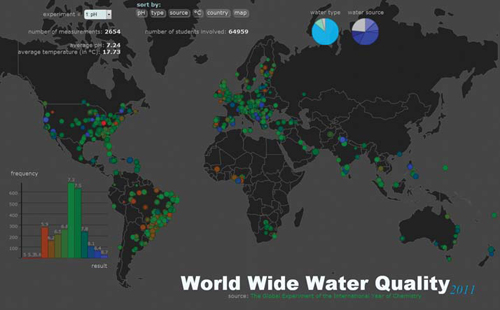 |
| Special software was developed for the Global Experiment to enable viewing, comparison, and analysis of the results. The program was created collaboratively by programmers around the world through a competition organized by Visualizing.org. |
In gratitude to the thousands of people who made the Global Experiment possible, IUPAC and UNESCO have released a video of this activity (produced by Casual Films).6 The video, which explains the objectives, the experiments, and the main conclusions of this activity, is a tribute to the thousands of students who collected and analyzed local water samples and shared their results online.
The year-long Global Experiment—Water: A Chemical Solution has been quite remarkable both educationally and socially. All of us in IUPAC and UNESCO are very grateful to everyone from around the world, who worked so hard to make it possible, but most importantly, we’d like to thank all the teachers and students who embraced the Global Experiment—they were the ones who made it truly global and made it such a success.
Javier Garcia Martinez <[email protected]> is a professor at the University of Alicante, a member of the IUPAC Bureau and the coordinator of the Global Experiment of the International Year of Chemistry. Rovani Sigamoney, <[email protected]> is UNESCO representative for IYC, from the UNESCO Division of Basic and Engineering Science. The Global Experiment executive team also included Colin Humphris (UK), Mark Cesa (USA), Peter Mahaffy (Canada), and Tony Wright (Australia). Several members from the IUPAC Committee on Chemistry Education and of the Analytical Chemistry Division were also involved, especially at the beginning during the experiments development phase; the group is presented in ref [2]; their valuable contribution is greatly acknowledged.
References
- IYC 2011 website: www.chemistry2011.org
- Tony Wright and Javier Garcia Martinez, “Water: A Chemical Solution—A Global Experiment for the International Year of Chemistry,” Chemistry International, Sep-Oct 2010, Vol. 32 No. 5, pp 14–17.
- The Global Experiment website: water.chemistry2011.org
Javier Garcia-Martinez, “Chemistry 2.0: Creating Online Communities,” Chemistry International, Jul-Aug 2010, Vol. 32, No. 4, pp. 4–7.
- Visualization Sprint: www.visualizing.org/sprint/global-water-experiment or www.chemistry2011.org/about-iyc/news/GWE_sprint_visualization
- Video release: http://youtu.be/mcKb9J0ZgxA or www.chemistry2011.org/participate/featured-ideas/be-inspired/
Page
last modified 30 April 2012.
Copyright © 2003-2012 International Union of Pure and Applied Chemistry.
Questions regarding the website, please contact [email protected] |