|
|
Vol.
33 No. 6
November-December 2011
| The Project Place |
| |
Information about new, current, and complete IUPAC projects and related initiatives.
See also www.iupac.org/projects |
The isotopic abundances (and atomic weights) of numerous elements (e.g., H, B, Li, C, N, O, Si, S, Ca, Fe, and Tl) vary in naturally occurring substances because molecules, atoms, and ions having different stable isotopes of the same chemical element possess slightly different physical and chemical properties. These elements commonly will be fractionated during physical, chemical, and biological processes, giving rise to variations in isotopic composition. Increasingly these variations in isotopic composition are being used in anthropology, atmospheric sciences, biology, chemistry, environmental sciences, food and drug authentication, forensic science, geochemistry, geology, medicine, oceanography, and paleoclimatology.
 |
 |
IUPAC’s Commission on Isotopic Abundances and Atomic Weights recognized that there was a lack of consensus on how to conduct such mass spectrometric measurements and how to express measurement results in stable isotope studies. Variations in isotopic compositions are relatively small. Typically, the difference in the ratio of the number of two stable isotopes in a specimen is compared with that in an internationally agreed reference material, and the result is reported as a delta value (δ). These values commonly are expressed in part per thousand (per mil, with symbol ‰). To improve mass spectrometric measurements and expression of their results, a project on guidelines for mass spectrometric isotope ratio measurements (2001-019-2-200) was begun in 2002. Under this project, papers have been published on mass spectrometric correction procedures and on recommendations for the inclusion of systematic errors in the publication of such measurements. The final paper in support of this project has been published in Rapid Communications and Mass Spectrometry titled “Guidelines and recommended terms for expression of stable-isotope-ratio and gas-ratio measurement results.”1 These guidelines:
- Are based on publications of the Commission on Isotopic Abundances and Atomic Weights, with input from more than 30 subject-matter experts.
- Provide examples of stable isotope measurement results consistent with the Système International d’Unités, the SI (known in English as the International System of Units), and the third edition of the International Vocabulary of Basic and General Terms in Metrology (VIM, 3rd edition).
- Encourage the use of and provide examples of stable-isotope-delta definition equations that are coherent quantity equations, where the extraneous factors 100, 1000, and 1 000 000 have been removed.
- Recommend and provide examples for expression of stable isotope and gas-ratio measurements using the symbols for dimensionless quantities, per mil (‰), part per million (ppm), and per meg (one part in a million parts), while recommending that mmol/mol (and similar units) be deprecated for expressing values of δ.
- Provide examples of column headings and axis labels for stable isotope and gas-ratio measurements consistent with quantity calculus.
- Recommend the analysis of international distributed isotopic reference materials along with sample unknowns and the publication of measurement results of the reference materials with sample unknowns.
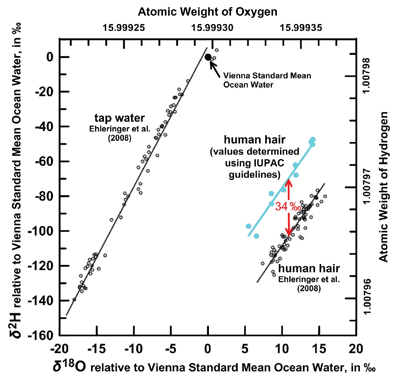 |
| Figure 1. Hydrogen isotopic composition (δ2H) versus oxygen isotopic composition (δ18O) of tap water and human scalp hair relative to Vienna Standard Mean Ocean Water. Open black circles are from Ehleringer et al.,[2] d IUPAC guidelines published in 1994.[4] |
The application of these guidelines in forensic science is shown in Figure 1, which is a plot of the hydrogen isotopic composition versus oxygen isotopic composition of tap water and of human scalp hair, primarily in North America. The spatial variation in isotopic composition of tap water is well documented and is a function primarily of temperature of formation of precipitation in clouds. Thus, groundwater and surface water (and tap water) is a function of elevation, distance inland from an ocean, etc. Tap water at the lower right of the figure derives from high elevations (e.g., Rocky Mountains), whereas tap waters near the top of the figure are illustrative of more equatorial sources. This figure shows that the hydrogen and oxygen isotopic composition are strongly correlated in tap water and scalp hair. Correlations in isotopic composition between tap water and scalp hair are used in human migration studies, in anthropology, forensic science, and potentially, for geospatial tracing of individuals. A fundamental problem was that the original human hair measurements,2 shown in the lower right of Figure 1, were not provided with isotopic compositions of reference materials available to the scientific community and were not calibrated satisfactorily to the stable hydrogen and oxygen isotope scales. Subsequently, Coplen and Qi3 prepared human hair isotopic reference materials for international distribution and improved analytical techniques. Following IUPAC guidelines,1,4 the hydrogen isotopic composition of human hair, shown in cyan in Figure 1, was found to be on average about 34 ‰ more positive than that of the original measurements on human hair.2 A major conclusion of this study is that readers need to pay attention to the hydrogen and oxygen isotopic compositions of isotopic reference materials in publications, and they may need to adjust these measurement results in previous publications, as needed, to ensure that all results are on the same isotope scales.
For more information, contact Task Group Chair Thomas Walczyk
<[email protected]>.
www.iupac.org/web/ins/2001-019-2-200
References
- T.B. Coplen, Guidelines and recommended terms for expression of stable-isotope-ratio and gas-ratio measurement results, Rapid Commun. Mass Spectrom. 2011, 25, 2538–2560. http://dx.doi.org/10.1002/rcm.5129
- J.R. Ehleringer, G.J. Bowen, L.A. Chesson, A.G. West, D.W. Podlesak, T.E. Cerling, Hydrogen and oxygen isotope ratios in human hair are related to geography, Proc. Natl. Acad. Sci. USA 105 (2008) 2788–2793. http://dx.doi.org/10.1073/pnas.0712228105
- T.B. Coplen, H.P. Qi, USGS42 and USGS43: Human-hair stable hydrogen and oxygen isotopic reference materials and analytical methods for forensic science and implications for published measurement results, Forensic Science International, (in press). http://dx.doi.org/10.1016/j.forsciint.2011.07.035
- T.B. Coplen, Reporting of stable hydrogen, carbon, and oxygen isotopic abundances, Pure Appl. Chem., 66, 273–276 (1994). http://dx.doi.org/10.1351/pac199466020273
Page
last modified 17 July 2012.
Copyright © 2003-2012 International Union of Pure and Applied Chemistry.
Questions regarding the website, please contact [email protected] |