|
|
Vol.
29 No. 3
May-June 2007
Assuring
Quality of Analytical Measurement Results: The IUPAC Role
by
Ales Fajgelj
Over
the past 30 years the value of world trade has risen dramatically.
In 2005 it amounted to almost USD 17 trillion (trillion =
1012; see figure 1). A large proportion of this
trade is dependent upon chemical analyses, since food, pharmaceutical
products, medicines, ores, and chemical products in general
represent the largest groups of trading items. To gain acceptance
in the trading process, the quality of analytical measurement
results needs to be assured and demonstrated. The term
quality of analytical measurement results encompasses, among
others, comparability of analytical results, their accuracy,
reproducibility, metrological traceability, measurement uncertainty,
and more.
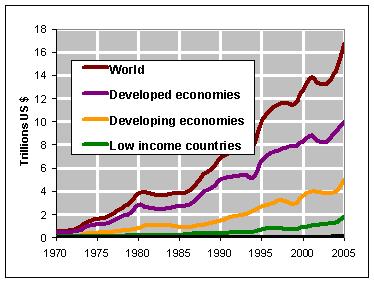
Figure
1: World Trade Development 1970–2005 in Trillions
of USD (Source: United Nations Conference on Trade and
Development).
IUPAC
has a long tradition of activities related to quality assurance
of analytical measurement results. The formation of the IUPAC/ISO/AOAC
Working Party for Harmonization of Quality Assurance Schemes
in 1978 was an important milestone. At that time, efforts
were focused on harmonizing requirements related to method
validation studies (or laboratory collaborative studies),
which had been conducted by a number of organizations around
the world. IUPAC, offering a completely neutral scientific
forum for harmonization activities, was identified as the
most appropriate body to host the working party. Today, after
almost 30 years, that working party is the IUPAC Interdivisional
Working Party for Harmonization of Quality Assurance (WPHQA),
which is part of the Analytical Chemistry Division (ACD).
The short description of activities that follows and the documents
cited here are aimed at highlighting the important role that
IUPAC, and specifically the WPHQA, plays in ensuring the quality
of analytical measurement results.
Method
Validation
The use of standardized methods of analysis in analytical
chemistry is one of the most traditional ways of achieving
comparability of measurement results. Especially in food analysis,
agrochemicals, organic analysis, and other analytical areas
where unstable samples and/or measurands are analyzed, the
use of standardized methods is often prescribed by legislation.
Two IUPAC internationally harmonized protocols have for many
years served as a basis for validation and adoption of standardized
analytical methods (procedures). The first is the IUPAC “Protocol
for the Design, Conduct, and Interpretation of Collaborative
Studies,”1 and the second
the “Harmonized Protocols for the Adoption of Standardized
Analytical Methods and for the Presentation of their Performance
Characteristics.”2 These
principles of collaborative studies for method validation
are still widely applied by the AOAC International, as well
as by the International Standards Organization (ISO). However,
the world is changing rapidly and with the fast development
of analytical instrumentation and the availability of new
analytical techniques and procedures the prescription of methods
to be used is sometimes a limiting factor. Responding to the
situation, the WPHQA has opened the door for single-laboratory
method validation, also known as in-house method validation.
The principles presented in the IUPAC “Harmonized Guidelines
for Single Laboratory Validation of Methods of Analysis”3
and in the proceedings of the Joint AOAC Int./FAO/IAEA/IUPAC
International Workshop on the Principles and Practices
of Method Validation, held in 1999 in Budapest, Hungary,4
have been accepted as official guidelines by the CODEX Alimentarius
Commission.
Quality
Control and Proficiency Testing
Established internal quality-control practices and regular
laboratory participation in proficiency testing constitute
another very important pillar of quality assurance in analytical
chemistry. Again, the contributions of the WPHQA have been
indispensable. Two IUPAC internationally harmonized documents,
namely the “International Harmonized Protocol for the
Proficiency Testing of (Chemical) Analytical Laboratories”5
and the “Harmonized Guidelines for Internal Quality
Control in Analytical Chemistry Laboratories”6
still provide the basic rules, which have received wide international
acceptance and utilization.
Assessment of laboratory performance based on a z-score evaluation
introduced in the IUPAC proficiency testing protocol became
the most frequently used approach in evaluation of laboratory
performance. Considering the experience gained over 13 years,
the protocol has been updated and a revised version titled
“The International Harmonized Protocol for the Proficiency
Testing (PT) of Analytical Chemistry Laboratories” was
published in 2006.7 To supplement
this so-called classical PT approach, the WPHQA recently initiated
a separate project on the Selection and Use of Proficiency
Testing Schemes for Limited Number of Participants (Chemical
Analytical Laboratories).8
In case of a small number of participants, some limitations
on statistical applications may appear and this project is
aimed at elaborating some additional approaches for evaluation
of participants’ results and their reporting.
However, neither of the above described external quality assurance
schemes replaces the internal laboratory quality control.
They actually should go hand in hand. The ISO/IEC 17025 standard,
which serves as a basis for laboratory accreditation, is very
general and brief in its Clause 5.9 titled “Assuring
the Quality of Test and Calibration Results.”9
It urges laboratories and accreditation bodies to use separate
guidance, specifically prepared for their field of application.
The large number of citations in scientific literature and
translations of both IUPAC documents into numerous languages,
including the translation by the Japan Chemical Laboratory
Accreditation into Japanese in 2001, reflect the importance
of this IUPAC activity for quality in chemical analytical
laboratories.
Metrological Traceability and Recovery
One of the most important parameters defining the quality
of analytical measurement results is comparability. Comparability
of measurement results is based on metrological traceability,
which allows results to be compared independently of the time,
place, analyst, and procedure used. Two aspects of this description
are very much IUPAC’s concern. The first is the metrological
traceability of chemical measurement results. It is a term
often used and cited, but without a firm agreement within
the measurement/scientific community regarding associated
concepts, their understanding, and requirements. In recent
years, IUPAC representatives have been deeply involved in
the ongoing revision of the International Vocabulary of
Basic and General Terms in Metrology10,
trying their best to assure that specifics of chemical measurements
are considered in this guide.

Comparability
of measurement is the ultimate goal of quality assurance
and is a prerequisite for smooth trade at the national,
regional, and global level.
In
addition, the WPHQA-coordinated project Metrological Traceability
of Measurement Results in Chemistry11
is also dealing with this issue. Concepts developed in the
framework of this project will be underpinned with examples
(various scenarios) for establishing traceability in chemical
measurement, to provide clear and practical explanations for
all levels of laboratories—from field laboratories to
metrology institutes. The guide will clarify terms like metrological
reference, traceability chain, and metrological hierarchy,
and describe the different roles that organizations in the
global metrological infrastructure (metrology institutes,
reference material producers, laboratories, etc.) have in
establishing metrological traceability. The guide is to be
presented during the IUPAC General Assembly in Torino, Italy,
in August 2007.
In discussions about metrological traceability of measurement
results one frequently hears the claim that the traceability
chain in chemistry has been broken. This claim is often related
to the chemical process, destructive analysis, where the sample
and measurand are converted into the physical and chemical
form suitable for the selected measurement technique/instrument.
Such conversions (digestions, extractions, etc.) may result
in the loss of measurand, incomplete conversion into the required
chemical/physical form, or even contamination, and are very
much dependent on the procedure used.
In
the context of this article, recovery is defined as the proportion
of the amount of analyte, present or added to the analytical
portion of the test material, which is extracted and presented
for measurement. It can be illustrated with the practical
example of the determination of pesticide residues in food.
The amount of the extracted, and consequently measured, pesticide
residue will depend on the procedure used. In the discussion
above, the use of standardized methods has been identified
as a possible solution to the problem. However, this is only
part of the overall process assuring the comparability of
measurement results. There is also different legislation in
different regions of the world. The European legislation in
this specific case requires reporting of results corrected
for recovery; this is not the case in the USA. It was a major
IUPAC success when IUPAC, ISO, and AOAC Int. agreed on the
technical principles for recovery determination provided in
the “Harmonized Guidelines for the Use of Recovery Information
in Analytical Measurement,”12
including the fact that recovery values need to be established
as a part of the method validation process and be available
if necessary, whether or not recoveries are reported or results
are corrected. On this basis measured values can always be
converted to corrected values and vice versa, thus enabling
comparability of results on a global scale.
 |
Combining
and Reporting Analytical Results
The correction of results for recovery, or not, is only one
illustrative problem related to reporting of analytical results.
There are many more. Combining measurement results obtained
by one analyst in one laboratory employing one measurement
procedure, and using one measurement technique is the starting
point for the two questions: How to report the associated
measurement uncertainty? and How to establish and demonstrate
the metrological traceability of combined results? The complexity
of these questions expands with the increasing number of measurement
procedures/techniques, and with the number of laboratories
and measurement results that need to be considered.
Continuing the tradition of organizing workshops and symposia,
the WPHQA, together with the Italian Agency for Environmental
Protection and Technical Services, organized the international
workshop Combining and Reporting Analytical Results—The
Role of (metrological) Traceability and (measurement) Uncertainty
for Comparing Analytical Results, in March 2006 in Rome, Italy.
The wide international interest in the topics covered by the
workshop program was reflected in the number of cosponsoring
organizations, namely: Centro Svillupo Materiali. S.p.A, the
International Atomic Energy Agency, the Consultative Committee
for Amount of Substance—Metrology in Chemistry (CCQM),
International Bureau of Weights and Measures, the Co-operation
on International Traceability in Analytical Chemistry, the
ISO Committee on Reference Materials and the United Nations
Industrial Development Organization.
Most lectures were prepared as full text for the proceedings
book published by the Royal Society of Chemistry.13
Contributions provide an overview of current practices used
in different laboratories from different scientific fields
to combine and report measurement results, at the same time
they describe some basic scientific considerations as well
as discussions related to legislative aspects. Practical examples
from environmental monitoring laboratories, reference material
producers, clinical chemistry, and the top metrological level
are included.
Although the workshop was a successful event, it represented
only one small step forward in providing answers for dilemmas
analytical chemists face in combining and reporting analytical
results. Much still needs to be done. In April 2007 a workshop
was organized by CCQM focusing on calculating the CCQM Key
Comparison Reference Values.
Harmonization
Today
From this article, the reader should recognize that in its
harmonization efforts IUPAC has never been working alone.
There has always been cooperation with relevant bodies and
other organizations. Cooperation is considered the only possible
approach to achieving agreement at a global level. However,
in the 1970s and early 1980s, the international standardization
and harmonization scene was smaller than today. Cooperation
between AOAC Int., ISO, and IUPAC was sufficient for assuring
appropriate arrangements and the flow of information. The
situation has changed drastically in the last 20 years. Metrology,
accreditation, and standardization infrastructures have developed
at all levels.
A careful look into the distribution of the most influential
organizations and bodies related to standardization and harmonization
in the area of analytical chemistry reveals that there is
a strong concentration in the northern hemisphere. The fact
is that barriers of trade exist and are still growing between
developed and developing economies. One reason for this is
the standardization and application of very strong quality
requirements in the accreditation process, without provision
of the required assistance and support to developing countries.
In this respect, the role of independent, non-commercial,
non-profit scientific organizations like IUPAC is of utmost
importance. The second important way of overcoming such differences
is by open access to scientific literature (e.g., via the
Internet). The IUPAC journal Pure and Applied Chemistry
is a valuable example.
References
1. W. Horowitz (1988), Pure Appl. Chem.
60(6), 855–864. (link
to TOC to access full text pdf)
2. W.D. Pocklington (1990), Pure Appl.
Chem. 62(1), 149–162. (link
to TOC to access full text pdf)
3. M. Thompson, S.R.L. Ellison, R. Wood
(2002), Pure
Appl. Chem. 74(5),
838–855.
4. A. Fajgelj, A. Ambrus, editors, (2000)
Principles and Practices of Method Validation. The
Royal Society of Chemistry, Special Publication No. 256, Cambridge
(ISBN 0-85404-783-2).
5. M. Thompson, R. Wood (1993), Pure
Appl. Chem. 65(9), 2123–2144.
(link
to TOC to access full text pdf)
6. M. Thompson, R. Wood (1995), Pure
Appl. Chem. 67(4), 649–666. (link
to TOC to access full text pdf)
7. M. Thompson, S.R.L. Ellison, R. Wood
(2006), Pure
Appl. Chem. 78(1),
145–196.
8. www.iupac.org/projects/2005/2005-019-2-500.html
9. ISO/IEC 17025 International Standard:
General Requirements for the Competence of Testing and Calibration
Laboratories (2005) International Organization for Standardization,
Geneva (ICS 03. 120.20).
10. BIPM, IEC, IFCC, ISO, IUPAC, IUPAP,
OIML (1993) “International Vocabulary of Basic and General
Terms in Metrology”, International Organization for
Standardization, Geneva (ISBN-92-67-01075-1), Second edition,
under revision.
11. www.iupac.org/projects/2001/2001-010-3-500.html
12. M. Thompson, S.R.L. Ellison, A. Fajgelj,
P. Willetts, R. Wood (1999), Pure Appl. Chem. 71(2),
337–348. (link
to TOC to access full text pdf)
13. A. Fajgelj, M. Belli, U. Sansone,
editors, Combining and Reporting Analytical Results
(2006) The Royal Society of Chemistry, Special Publication
No. 307, Cambridge (ISBN-13 978-0-85404-848-9) <www.rsc.org/Publishing/Books/0854048480.asp>
A.
Fajgelj <[email protected]>
works at the International Atomic Energy Agency in Vienna,
Austria. In IUPAC, he is vice president of the Analytical
Chemistry Divsion, chairman of the Interdivisional Working
Party for Harmonization of Quality Assurance, and a member
of the Subcommittee on Food Chemistry of the Chemistry and
the Environment Division. He is also the IUPAC representative
on the International Committee on Weights and Measures/Consultative
Committee on Amount of Substance and on the ISO—Committee
on Reference Materials.
www.iupac.org/divisions/V/501
Page
last modified 15 June 2007.
Copyright © 2003-2007 International Union of Pure and
Applied Chemistry.
Questions regarding the website, please contact [email protected]
|