|
|
Vol.
28 No. 3
May-June 2006
Special
Topic Articles Featuring the 2005 Winners of the IUPAC Prizes
for Young Chemists
Pure and Applied Chemistry
Vol.
78, No. 1, pp. 1–64, 2006
As
an international nongovernmental scientific organization,
IUPAC takes great interest in the worldwide achievements of
chemists, particularly young chemists. Therefore, IUPAC established
the annual prestigious Prizes for Young Chemists. In doing
so, we endeavor to encourage research in the chemical sciences
and the participation of promising young chemists.
Starting in 2002, prizewinners have been invited to submit
manuscripts on aspects of their research topics for consideration
as short, critical review articles to be published in
Pure and Applied Chemistry. Following peer review, the
first collection appeared in PAC 74(11), 2021–2081
(2002) and encouraged the view that it offers sufficient readership
appeal to become a regular special topic feature of the journal.
The second series, covering the works of the 2003 winners
was published in PAC 76(2), 263–319 (2004),
and the third series in PAC 76(12), 2051–2099
(2004). The most recent series of articles was published in
the January 2006 issue of PAC and includes the following
critical reviews:
“Evolutionary
Approaches for the Discovery of Functional Synthetic Small
Molecules,” by Zev J. Gartner (pp. 1-14)
Directed evolution is a powerful method for the laboratory
discovery of nucleic acids and proteins with desired functional
properties. A hallmark of this approach is the iterative translation,
selection, amplification, and diversification of genetic information.
The potential of evolutionary methods to impact the discovery
of synthetic small molecules has recently been explored by
a variety of laboratories. Four methods encompassing some
or all of the hallmarks of evolution are discussed, including
dynamic combinatorial chemistry, genetic algorithms, DNA display,
and DNA-templated synthesis.
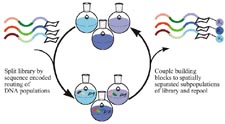
DNA
display: DNA libraries are split into subpopulations according
to their sequences. Each subpopulation, having in common
a particular DNA codon, is subjected to a particular set
of reaction conditions. The reacted subpopulations are
re-pooled and subjected to further manipulations. (by
Z.J. Gartner)
“Syntheses
and Applications of Conducting Polymer Polyaniline Nanofibers,”
by Jiaxing Huang (pp. 15-27)
Nanofibers with diameters of tens of nanometers appear to
be an intrinsic morphological unit that was found to "naturally"
form in the early stage of the chemical oxidative polymerization
of aniline. In conventional polymerization, nanofibers are
subject to secondary growth of irregularly shaped particles,
which leads to the final granular agglomerates. The key to
producing pure nanofibers is to suppress secondary growth.
Based on this, two methods—interfacial polymerization
and rapidly mixed reactions—have been developed that
can readily produce pure nanofibers by slightly modifying
the conventional chemical synthesis of polyaniline without
the need for any template or structural directing material.
With this nanofiber morphology, the dispersibility and processibility
of polyaniline are now much improved. The nanofibers show
dramatically enhanced performance over conventional polyaniline
applications such as in chemical sensors. They can also serve
as a template to grow inorganic/polyaniline nanocomposites
that lead to exciting properties such as electrical bistability
that can be used for nonvolatile memory devices. Additionally,
a novel flash welding technique for the nanofibers has been
developed that can be used to make asymmetric polymer membranes,
form patterned nanofiber films, and create polymer-based nanocomposites
based on an enhanced photothermal effect observed in these
highly conjugated polymeric nanofibers.
 |
Flash
welding technique can be used to create patterns in nanofiber
films. The top optical microscopy image shows a copper
grid mask lying on top of a polyaniline nanofiber film.
After exposure to a camera flash, the grid pattern is
generated on the nanofiber film. The unmasked diamond
shaped areas are welded, therefore, reflect more light
and look bright under an optical microscope. The previously
masked areas still look green (scale bar:100 m). (by J.
Huang) |
“A
Dozen Years of N-Confusion: From Synthesis to Supramolecular
Chemistry,”
by Hiromitsu Maeda and Hiroyuki Furuta (pp. 29-44)
The chemistry of N-confused porphyrin (NCP) and its analogs
started in 1994. Since then, considerable progress has been
made in understanding the unique properties of NCP and its
analogs, which confer characteristic reactivity and metal
complex formation. The evolved isomers, multiply NCPs, and
expanded N-confused derivatives, have opened up new realms
of NCP chemistry. Cis- and trans-doubly
N-confused porphyrin (N2CP) stabilizes higher oxidation
states such as CuIII in square-planar fashion in
the core. Confused isomers with five or more pyrrole rings
can coordinate several cations owing to their larger cavities
compared to tetrapyrrolic system. The peripheral nitrogen(s)
of NCP and its analogs can serve as hydrogen-bonding donor
and acceptor, and metal coordination site as well. For example,
NCP forms versatile dimers with the assistance of metal ions.
The square-planar divalent metal complexes of C6F5-substituted
NCP act as efficient anion-binding receptors. Furthermore,
CuIII complexes of N2CP, possessing
both N and NH at the periphery, form self-assembled one-dimensional
(1D) hydrogen-bonding networks, whose orientations differ
in cis (zigzag) and trans (straight) isomers.
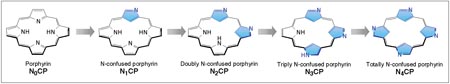
Evolution
of the N-confused porphyrin (NCP) family. (by H. Maeda)
“Solution-Based
Routes to Transition-Metal Oxide One-Dimensional Nanostructures,”
by Xun Wang and Yadong Li (pp. 45-64)
One-dimensional (1D) nanostructures have drawn continuous
research attention because of their unique electrical, optical,
and magnetic properties different from that of bulk and nanoparticles,
as well as their potential applications in mesoscopic research
and nanodevices. The main challenge in this area is how to
precisely control the sizes, dimensionalities, compositions,
and crystal structures in nanoscale, which may serve as a
powerful tool for the tailoring of physical/chemical properties
of materials in a controllable way. Here, we review the advances
in the solution-based routes to prepare 1D nanostructures.
Particularly, three systems of MnO2, rare-earth
compounds, and silicates have been chosen to show the synthetic
strategy under hydrothermal conditions. As the main theme,
a rolling mechanism has been given special attention to present
a relative general understanding of the growth of various
transition-metal oxide 1D nanostructures under solution conditions.
| TEM
image of δ-MnO2
nanotubes
with α-NaMnO2
as precursors. (Xun Wang) |
 |
www.iupac.org/publications/pac/2006/7801
Page
last modified 25 April 2007.
Copyright © 2003-2007 International Union of Pure and
Applied Chemistry.
Questions regarding the website, please contact [email protected]
|